|
1/13/2024 0 Comments Hollow cathode lamp block diagram 
The hollow cathode lamp the primary requirement of a hollow cathode lamp is to gen erate a narrow emission line of the element which is being measured. There is a glow discharge between the cathode and the anode in which positive gas ions are formed which sputter element atoms of the cathode at relatively low temperatures. Basic diagram of a hollow cathode lamp hollow cathode lamps from an atomic absorption spectrometer a hollow cathode lamp hcl is type of cold cathode lamp used in physics and chemistry as a spectral line source e g. This line should be of sufficient spectral purity and intensity to achieve a linear calibration graph with low noise level from the aa spectrometer. The glass tube is filled with some type of inert gas such as argon or neon with a pressure of around 5 torr 666 pascal. The key requirement for the hollow cathode lamp is to generate. 
Hollow cathode lamps are ideal source of spectral lines that are used for testing in laboratories. Explain how the following instrumental components work, in detail, with the aid of diagrams: 12 points) (a) Silicon Photodiode (b) Hollow Cathode Lamp (c) Photomultiplier Tube. It is important that the gas is inert in order to minimize interferences in the output of the data. This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. Diagram of a hollow cathode lamp a hcl consists of an anode and a cathode inside of a glass tube. Hollow cathode effect was first observed in 1916 by friedrich paschen a german physicist known for his work on electrical discharges. A single or multi element hollow cathode lamp is required for each element to be determined using the aa technique. Hollow cathode lamp the hollow cathode lamp hcl uses a cathode made of the element of interest with a low internal pressure of an inert gas. The hollow cathode lamp is a discharge lamp designed for use as a spectral line source with atomic absorption aa spectrometers. Two types of light sources are mostly used. For atomic absorption spectrometers and as a frequency tuner for light sources such as lasers. 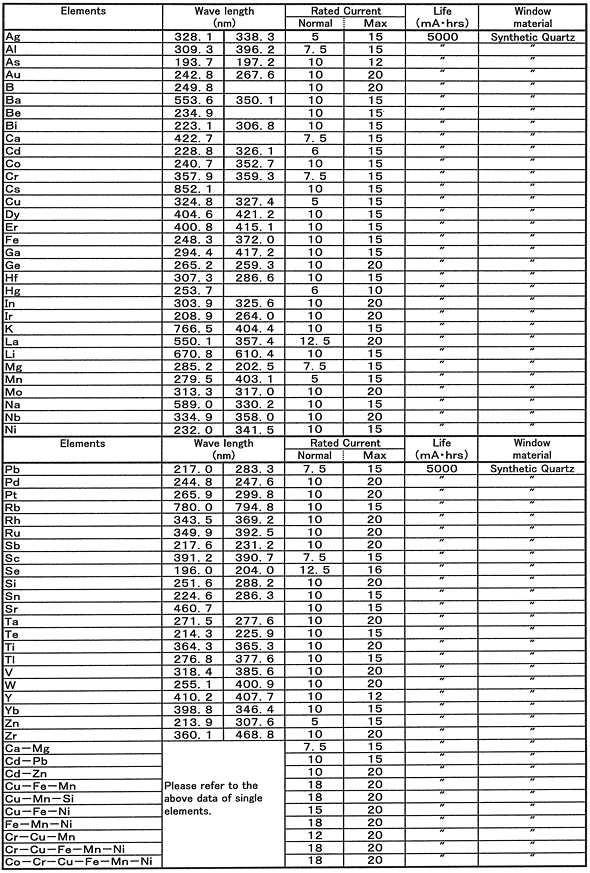
A typical hollow cathode lamp construction. Increasing the current too much can also result in a shorter lifetime of the lamp, as well as melting the metal on the cathode thus ruining the lamp.Hollow cathode lamp diagram. The process of self adsorption results in weaker intensities. ach component will be used once when labeling the diagram and once when matching to the description Anode Ne or Ar gas ransparent window Hollow cathode It is made of tungsten. Then match the component with its description. These unexcited atoms can interfere with good output data because they are able to absorb the radiation that is emitted from the excited electrons returning to their ground state. Science Chemistry Chemistry questions and answers el the components of the hollow-cathode lamp below. These higher currents allow for more intense data output but at the same time can produce a number of unexcited atoms in the cloud. High-intensity hollow cathode lamps (HI-HCLs. HCLs are almost ideal line sources for AAS because of their high stability and narrow linewidth (0.002 nm), but their relatively low intensity is a disadvantage for AFS. Then put in a small series of sentences to answer the question referring to the diagram. A HCL is composed of a silica envelope that contains 15 Torr of argon or neon and two metal electrodes. When a high voltage is applied, higher currents arise. How does a hollow cathode lamp deliver light of such a small bandwidth Youll need to draw a diagram here. How well the HCL works and how long it lasts is dependent on the care of operation. The material of the window is specifically selected in order to get the best transmission of spectral lines for the cathode element. Contamination of the cathode compromises the purity of the metal of interest and the data obtained for that metal. It is important that the cathode be stored under a vacuum to avoid any kind of contamination. The cathode is made of the pure metal of interest or a mixture of metals containing the metal of interest. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
